When a patient walks out with a prescription for generic drugs medications that contain the same active ingredient, strength, dosage form, and route of administration as a brand-name drug, approved by the FDA as therapeutically equivalent, they expect the same results. But what if the doctor prescribing it doesn’t fully believe in it? That’s the hidden problem behind low generic adoption - not patient resistance, but clinician doubt.
Despite generics making up 90% of all prescriptions in the U.S. and saving the healthcare system over $2 trillion in the last decade, many clinicians still hesitate. A 2017 study found that 68% of physicians had at least some concern about whether generics truly worked the same as brand-name drugs. That’s not because the science is shaky - it’s because education hasn’t kept up.
Why Clinicians Still Doubt Generics
The FDA requires generic drugs to meet strict bioequivalence standards. To be approved, a generic must show that its absorption in the body (measured by AUC and Cmax) falls within 80-125% of the brand-name drug. That’s not a wide range - it’s tight enough to guarantee identical therapeutic effect. Yet, a 2020 survey of 1,200 prescribers revealed that 45% incorrectly believed generics must have the same inactive ingredients. Another 38% thought manufacturing standards for generics were lower. And 27% wrongly assumed generics could contain up to 20% less active ingredient.
These aren’t minor misunderstandings. They’re foundational errors that shape prescribing habits. A 2019 analysis found that 62% of physicians still defaulted to brand names when writing prescriptions - even when the generic was available and cheaper. Why? Because they learned the brand name in medical school, heard it from senior doctors, and never got clear, practical training on the science behind generics.
The Real Cost of Misunderstanding
The consequences aren’t just financial. When clinicians don’t endorse generics, patients pick up on it. A Harvard Medical School study showed that when doctors said, “This generic is just as good,” patients reported 18% fewer side effects - even though the drug was identical. That’s not placebo; that’s nocebo. The fear of inferior quality triggers real physical symptoms.
And it affects adherence. Patients are 35% more likely to start and stick with a medication if it’s generic - not because it’s cheaper alone, but because they trust their provider’s recommendation. When a clinician hesitates, the patient hesitates too. In chronic conditions like hypertension or depression, that hesitation can mean hospitalization, worsening disease, or even death.
What Clinicians Actually Need to Know
Effective education doesn’t mean rehashing FDA regulations. It means answering the questions clinicians actually ask:
- Do inactive ingredients really matter? (Answer: Only if they cause an allergy. Otherwise, they’re harmless fillers.)
- Is there a difference in quality between generic manufacturers? (Answer: All must pass the same FDA inspections. Some are even made in the same factories as brand-name drugs.)
- Why do some patients say the generic “doesn’t work”? (Answer: Often it’s a nocebo effect - or a switching issue, not a drug issue.)
- How do I explain this to a skeptical patient? (Answer: Use simple language: “This pill has the same medicine as the brand, just without the fancy packaging.”)
One major gap is in interpreting the FDA’s Orange Book the official list of approved drug products with therapeutic equivalence evaluations, using ‘A’ and ‘B’ ratings to indicate equivalence. Clinicians need to know that an ‘A’ rating means interchangeable. An ‘B’ rating means it’s not. Most don’t check it.

What Works: Real-World Education That Changes Behavior
Passive materials - PDFs, brochures, webinars - barely move the needle. A 2021 study in JAMA Internal Medicine found that clinicians who went through interactive, case-based training retained 42% more knowledge six months later than those who just read handouts.
Successful programs do three things:
- They embed learning into real workflows. For example, the University of California San Francisco added a pop-up alert in their EHR system: “This patient is on brand-name atorvastatin. Generic is available and costs $12 less per month. Would you like to switch?”
- They use spaced repetition. One program gave clinicians four 90-minute sessions over six months. Retention jumped 52% compared to a single one-hour lecture.
- They target specialties with high resistance. Cardiologists and neurologists are the most skeptical. Tailored education for them cut brand-name prescribing by over 30% in pilot programs.
Even simple changes help. A 2023 pilot with Medisafe’s AI tool showed that when EHR systems flagged generic alternatives at the point of prescribing, generic acceptance rose by 24%.
Where Education Falls Short
Not all efforts succeed. A 2020 Medicaid program in Tennessee spent $1.2 million on physician education - and saw only an 8% increase in generic use. Why? The materials weren’t integrated into the EHR. Doctors didn’t see them during patient visits. They were just another email to ignore.
Another blind spot is biosimilars. Many clinicians still confuse them with generics. Biosimilars complex biologic drugs that are highly similar to an approved reference product, but not identical due to their large-molecule structure are not the same as small-molecule generics. Only 31% of providers could correctly explain the difference in a 2023 FDA survey. That’s dangerous - especially in oncology, where missteps can cost lives.
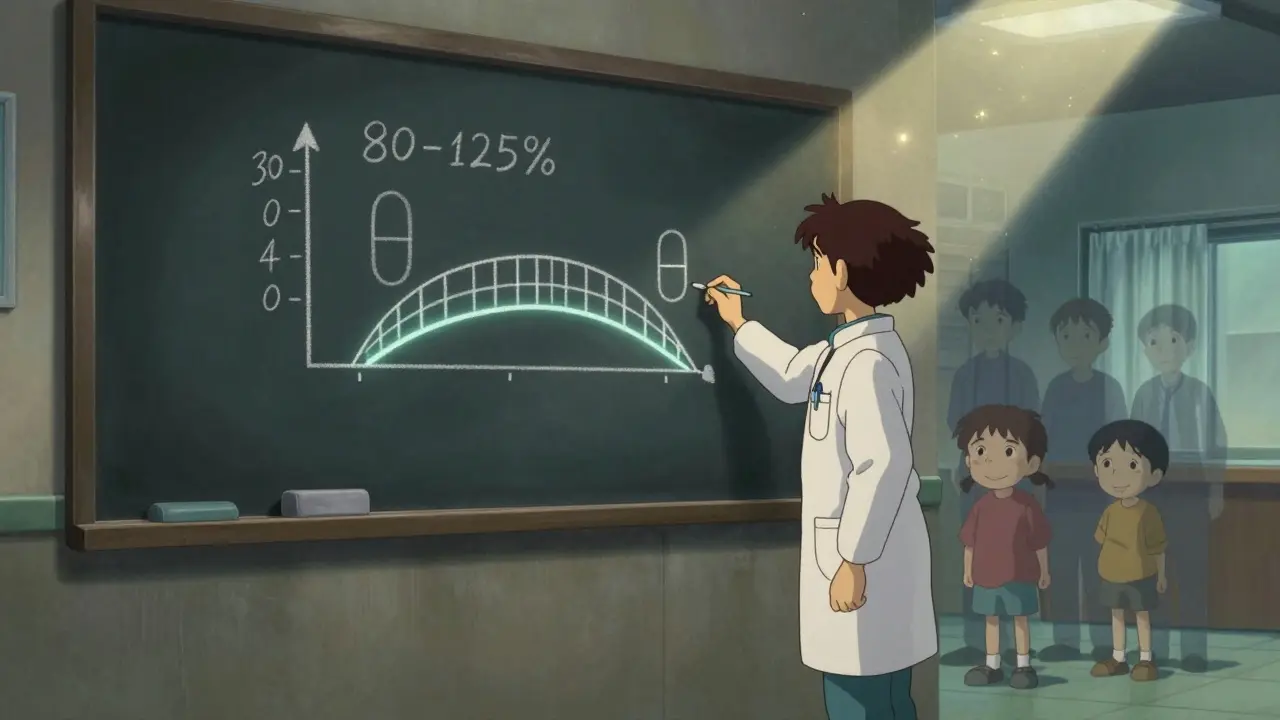
How to Get Started
You don’t need a big budget. Start here:
- Download the FDA’s Generic Drug Facts Handout - it’s free, clear, and designed for clinicians.
- Use the Orange Book to check therapeutic equivalence before prescribing.
- Ask your pharmacy: “What’s the cost difference between this brand and its generic?” Use that number in conversations.
- Start one conversation a day with a patient: “I know you’ve heard mixed things about generics. Let me explain why this one is safe and effective.”
And if your clinic or hospital doesn’t offer training, ask for it. A 2022 poll of 4,200 physicians showed 63% wanted more continuing education on generics. You’re not alone.
The Bigger Picture
The future of healthcare isn’t just about better drugs - it’s about better prescribing. The Centers for Medicare & Medicaid Services plans to include generic prescribing rates in its 2025 Merit-based Incentive Payment System (MIPS). That means your prescribing habits could affect your reimbursement.
But beyond incentives, there’s ethics. Every time you default to a brand-name drug without reason, you’re costing the patient - and the system - money. And every time you confidently prescribe a generic, you’re giving someone a chance to stay healthy.
Generics aren’t second-rate. They’re science-backed, cost-effective, and just as safe. The problem isn’t the drug. It’s the knowledge gap. And that’s something you can fix - starting today.
Are generic drugs really as effective as brand-name drugs?
Yes. The FDA requires generics to have the same active ingredient, strength, dosage form, and route of administration as the brand-name drug. They must also prove bioequivalence - meaning their absorption in the body falls within 80-125% of the brand. This isn’t a rough estimate; it’s a strict scientific standard. Over 90% of prescriptions in the U.S. are for generics, and they’ve saved $2.2 trillion in the last decade - because they work.
Why do some patients say the generic doesn’t work for them?
Often, it’s not the drug - it’s the mind. Studies show that when patients believe a generic is inferior, they report more side effects - even when the drug is identical. This is called the nocebo effect. It’s why provider endorsement matters. If you say, “This generic is just as good,” patients are 18% less likely to report side effects. Switching from one generic to another can also cause temporary adjustment, but that’s not a sign of ineffectiveness.
Can pharmacists substitute generics without my permission?
It depends on the state. In 19 states, pharmacists can substitute a generic without telling you - unless you’ve written “dispense as written” on the prescription. In 16 states, they must get your approval first. The rest have mixed rules. Always check your state’s laws. But remember: if the drug is rated ‘A’ in the FDA’s Orange Book, substitution is safe and legal.
What’s the difference between a generic and a biosimilar?
Generics are chemically identical copies of small-molecule drugs - like metformin or lisinopril. Biosimilars are highly similar versions of complex biologic drugs - like insulin or Humira. Because biologics are made from living cells, they can’t be exact copies. Biosimilars must show no meaningful clinical difference, but they’re not considered interchangeable unless specifically approved. Only 31% of providers could correctly explain this in a 2023 FDA survey. Always check the label.
How can I learn more about generic drugs as a clinician?
Start with the FDA’s free resources: the Generic Drug Facts Handout and the Generic Drugs and Health Equity Handout. Both are designed for prescribers. Also check out the Generic Pharmaceutical Association’s educational modules. For deeper learning, look for case-based CME courses - they’re proven to improve retention. And don’t ignore your pharmacy team. Pharmacists are often the best source of real-time, practical advice on generic substitution.
Provider education on generics isn’t about pushing cheaper drugs - it’s about empowering clinicians with accurate science. When you know the facts, you can make better choices - and help your patients stay healthy without unnecessary cost.


Oh wow, another article telling doctors they’re dumb for not knowing generics are the same.
Let me guess-next you’ll tell us that water is wet and gravity isn’t just a suggestion.
Meanwhile, I’ve had patients crash because their ‘equivalent’ generic had a different filler that triggered an allergic reaction. Oh wait, that’s not in the FDA’s 80-125% math, is it?
Maybe instead of lecturing, someone should admit that ‘bioequivalence’ is a statistical loophole, not a medical guarantee.
I can't believe this. 😔
Doctors are being blamed for patients not trusting generics? What about the pharma companies that spent decades marketing brand names as 'superior'?
And now we're supposed to trust some pill with a different color and shape just because the FDA says so?
My grandma took a generic for years-then had a stroke. Coincidence? Or did the 'same active ingredient' not include the same quality control?
They're not equal. They're just cheaper. And we're pretending otherwise.
There’s a profound irony in how we treat medical knowledge.
We demand precision in surgery, accuracy in diagnostics, and rigorous evidence in trials-yet when it comes to generics, we treat the science like a suggestion.
The FDA’s bioequivalence standards aren’t arbitrary; they’re rooted in pharmacokinetic modeling, population variance, and decades of clinical outcomes.
The real issue isn’t ignorance-it’s cognitive dissonance. We’ve been conditioned to equate price with quality, and branding with trust.
Fixing this requires more than education. It requires a cultural reckoning with how we assign value to health.
The data is clear and overwhelming
Generics are therapeutically equivalent and cost effective
Physician education must be integrated into clinical workflow not treated as an afterthought
Continuing medical education should be mandatory and case based
The healthcare system cannot afford to delay this improvement any longer
Yeah sure, generics are fine until you’re the one on one that made you feel like you got poisoned.
And don’t even get me started on the fact that half of them are made overseas with ingredients that wouldn’t pass a Walmart quality check.
My cousin’s dad died because his blood pressure med was ‘equivalent’-turns out the batch had 12% less active ingredient.
So yeah, science says it’s fine. But science didn’t bury him.
The issue is not whether generics are scientifically equivalent, but whether the regulatory framework adequately accounts for inter-individual pharmacodynamic variability.
While AUC and Cmax thresholds are statistically valid, they do not capture subpopulations with altered metabolism, polypharmacy interactions, or genetic polymorphisms affecting drug transporters.
Furthermore, the Orange Book’s ‘A’ rating does not imply identical clinical outcomes across all demographics.
Education must address pharmacogenomics, not just FDA compliance.
I’ve been a pharmacist for 22 years and I can tell you this: 98% of the time, generics work exactly the same.
The 2% of cases where someone has a reaction? Usually it’s a filler, not the active ingredient.
And yes, some generics are made in the same factory as the brand. I’ve seen the invoices.
Stop listening to anecdotes. Look at the data.
When you switch 10,000 patients from brand to generic and monitor outcomes? Hospitalizations drop. Adherence goes up. Costs go down.
It’s not magic. It’s math.
I grew up in a small village in India where generics were the only option.
No one had brand names. No one had insurance.
But people lived. They managed diabetes. They controlled seizures. They raised kids.
Here in the U.S., we turn a science-backed, life-saving option into a philosophical debate.
Maybe the real problem isn’t what doctors don’t know-it’s what we’ve been taught to fear.